Diffusion
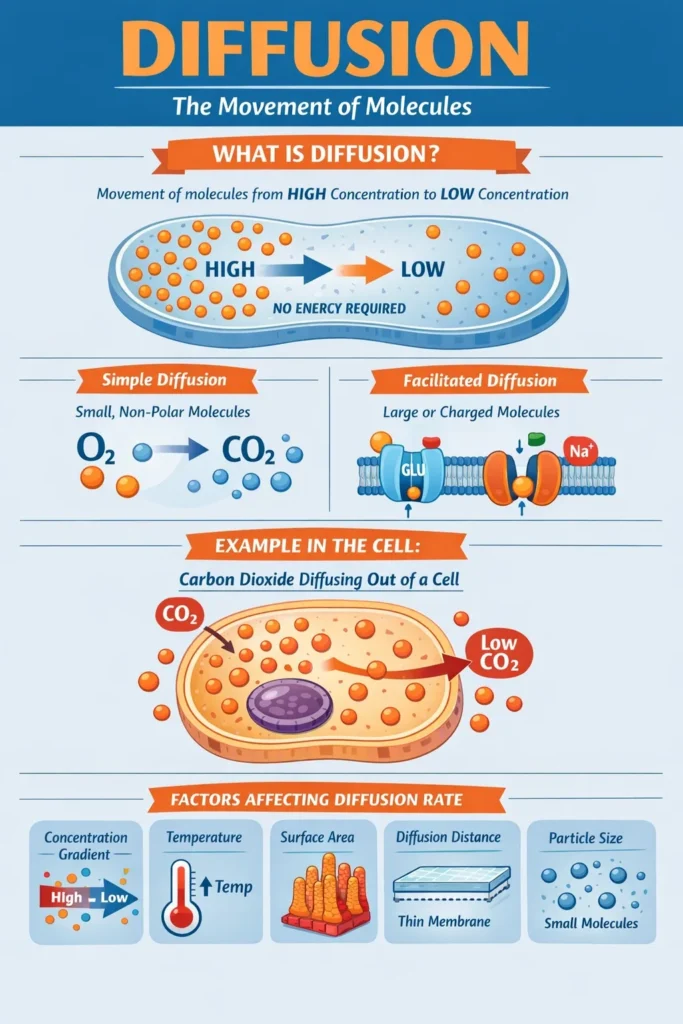
The transfer of molecules from a higher concentration to a lower concentration without the need for energy is known as diffusion. This can happen in the cell when substances migrate inside the cytosol or when tiny molecules diffuse past a cell membrane hole.
When a certain molecule is more concentrated in one place than another, diffusion takes place, creating a concentration gradient. One example of passive transport is the diffusion of molecules over a concentration gradient, which does not require a transport protein.
Molecules migrate from a higher to a lower concentration during diffusion until the concentrations in each location are equal. Diffusion will happen more quickly if the concentration gradient is greater and, if a barrier is present, it is permeable.
This kind of transport allows substances like gases and water to enter the cell. Imagine, for instance, that you drop a cube of sugar into a glass of water without stirring. Through the act of diffusion, the sugar will gradually dissolve and spread throughout the water.
During cell respiration, carbon dioxide is created within the cell. Diffusion is a necessary mechanism for cell respiration. A high concentration of carbon dioxide molecules accumulates inside the cell as a result of respiration. The above diagram shows that while molecules diffuse both into and out of the cell, the net (overall) migration is out of the cell and along the concentration gradient. We refer to this as the molecules trying to become isotonic. Diffusion is caused by the particles’ inherent kinetic energy; they don’t need an additional energy source from respiration.
Types of Diffusion in Biology
Two main forms of diffusion are used by biological systems to transfer materials across cell membranes:
Simple Diffusion
Small, non-polar molecules (such as carbon dioxide and oxygen) move across the cell membrane’s phospholipid bilayer on their own.
Facilitated Diffusion
Certain protein channels or carrier proteins implanted in the membrane allow larger or charged molecules (such as glucose or sodium ions) to pass through. Despite the need for these “helpers,” the process is still passive and follows the concentration gradient.
Factors Influencing the Diffusion Rate
Fick’s Law states that many factors influence the rate of diffusion:
- Concentration Gradient: A quicker rate results from a steeper gradient, or a greater concentration difference.
- Temperature: Particles travel and diffuse more quickly at higher temperatures because they have more kinetic energy.
- Surface Area: More room for particles to traverse concurrently is provided by a membrane with a bigger surface area, such as the gut’s microvilli.
- Diffusion Distance: Transport is accelerated by thinner membranes, such as the alveolar walls’ one-cell thickness.
- Particle Size: Compared to bigger, heavier particles, smaller, lighter particles disperse more quickly.
Interesting Examples of Diffusion
- During respiration, carbon dioxide diffuses from the blood into the lungs to be expelled, and oxygen diffuses from the air sacs in the lungs into the blood.
- Digestion: Food molecules that have been broken down in the small intestine, such as glucose, diffuse into the circulation.
- Everyday Life: Classic non-biological examples of dispersion include smelling perfume from across a room or seeing a tea bag color a cup of hot water.
Osmosis
The passage of water across a selectively permeable membrane due to a concentration gradient is known as osmosis. In other words, it is a unique kind of diffusion that is solely focused on water. The solute concentration is equalized across a permeable membrane as a result of the water moving in the direction of a high solute concentration.
From the perspective of the water, it diffuses, going from a high concentration to a lower concentration. Since osmosis doesn’t involve energy expenditure, it is also a type of passive transport.
The process of osmosis is crucial for transferring water between cells. Animal and plant cell membranes, including the inner membrane around a plant’s vacuole, are somewhat permeable. Visking tubing can be used in an experiment to demonstrate the osmosis process, even if we are unable to demonstrate the effects of osmosis in cells effectively.
Important Features of Osmosis
- Energy (ATP) is not needed for a passive process.
- Selective Barrier: The barrier prevents bigger solute particles like sugar or salt from passing through, allowing only the solvent (water) to do so.
- The objective is to keep going until balance is achieved, or when the water concentration is the same on both sides.
Effects on Cells (Tonicity)
How a cell responds to its environment depends on the concentration of the surrounding solution:
| Solution Type | Defination | Effect on Animal Cell | Effect on Plant Cell |
| Hypotonic | Lower solute outside | Swells and may burst (lysis) | Becomes turgid (firm); ideal state |
| Isotonic | Equal solute | Stays normal; ideal state | Becomes flaccid (soft/droopy) |
| Hypertonic | Higher solute outside | Shrivels (crenation) | Plasmolysis (membrane pulls from the wall) |
Biological Significance OF Osmosis
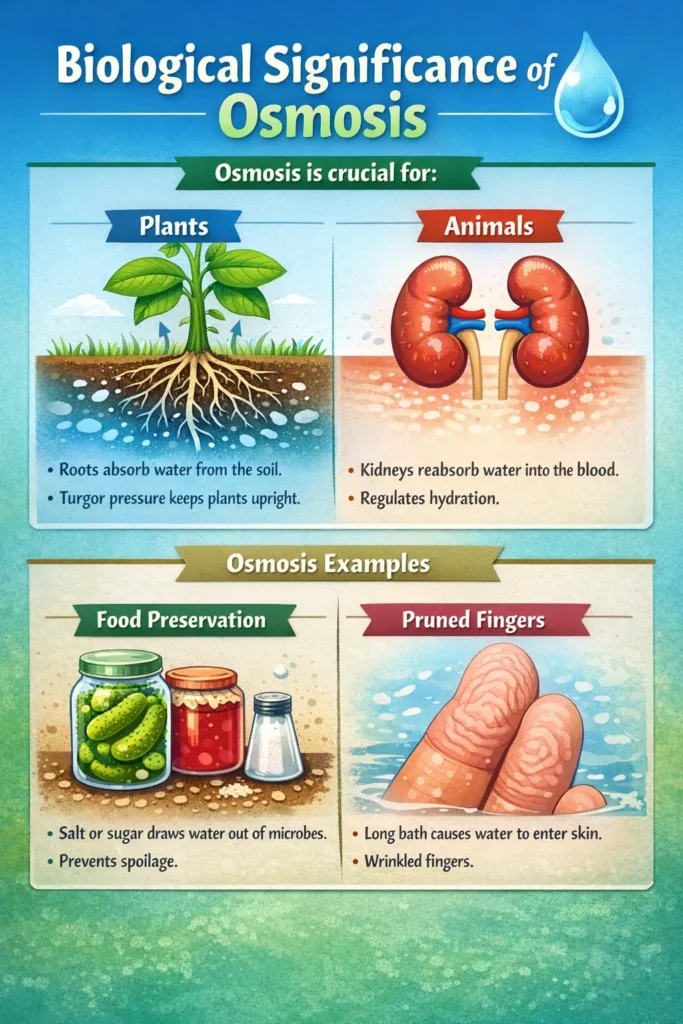
- Osmosis is the process by which roots of plants take up water from the soil. Turgor pressure is produced, keeping plants erect and preventing withering.
- In animals, it is essential for kidney function (reabsorbing water into the blood) and controls hydration.
Osmosis Examples
- Food Preservation: Environments with a lot of salt or sugar extract water from microorganisms, avoiding spoiling.
- Pruned Fingers: As water seeps into the cells during a lengthy bath, the skin wrinkles.
Active Transport
Moving substances against their concentration gradient, from a region of low concentration to one of high concentration, is known as active transport.
In contrast to diffusion and osmosis, this is an active process that requires energy from the cell.
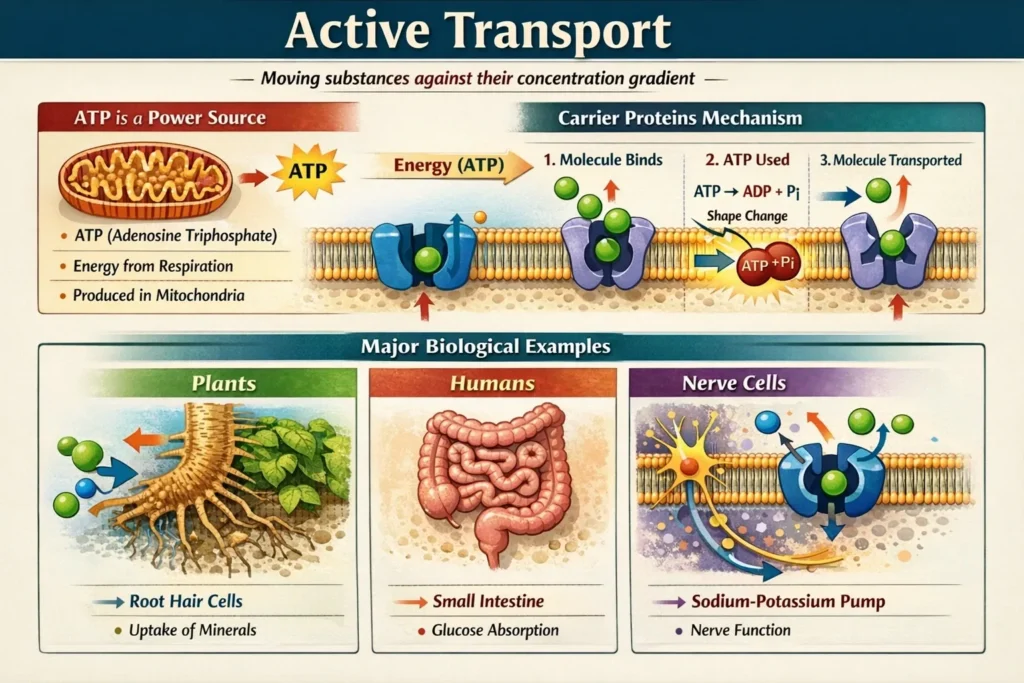
A transporter protein and an energy source are necessary for the active transport of molecules across a cell membrane. It differs from passive transport in that it requires energy. This activity is crucial for moving chemicals that are present in the extracellular environment at extremely low concentrations across the cell membrane.
ATP is a Power Source
- ATP (adenosine triphosphate) is the chemical energy needed for active transport.
- This energy is created in the mitochondria during respiration.
- Cells that perform a lot of active transport (like root hair cells or kidney tubule cells) usually have a high number of mitochondria.
Carrier Proteins Mechanism
- Specialized protein pumps or carrier proteins are found in the cell membrane.
- The molecule attaches itself to a particular location on the protein.
- The protein modifies its form using ATP-derived energy.
- This causes the molecule to be “pumped” across the membrane and released on the other side.
Major Biological Examples
- Plants (Root Hair Cells): The concentration of mineral ions, such as magnesium and nitrates, is often higher inside plants than in the soil. The roots may “pull” these minerals in against the gradient thanks to active transport.
- Humans (Small Intestine): Following a meal, glucose diffuses into the circulation. Active transport takes over to absorb the remaining glucose once the concentrations are equal, preventing nutritional loss.
- Nerve Cells: Our brain transmits electrical messages using the Sodium-Potassium Pump, which employs active transport to transfer ions into and out of neurons.